The Potential Role of Spa Therapy in Managing Frailty in Rheumatic Patients: A Scoping Review
Keywords
Article abstract
Frailty is not limited to the elderly, as patients with rheumatic diseases can also experience this condition. The present scoping review aimed to investigate the possibility of using the health resort setting as an alternative location for managing rheumatic patients with frailty. The research resulted in finding several in vitro, in vivo, and clinical studies, resulting in evidence supporting the effectiveness of spa treatments in reducing pain, improving function, and managing comorbidity in rheumatic diseases. Additionally, spa treatments were demonstrated to modulate the MAPK/ERK pathway and the NF-kB pathway's activation and to reduce proinflammatory molecules' secretion in rheumatic diseases, thus suggesting their potential effective role in the regulation of inflammaging in frailty. Moreover, the health resort setting may offer potential resources to reduce risk factors, such as drug consumption, inactivity, and disease severity, and may serve as a setting for developing prevention protocols for frailty. Future research should explore innovative approaches, such as exercise training and early diagnostics, for the overall management of frailty in rheumatic patients in the spa setting.
Article content
1. Introduction
2. Materials and Methods
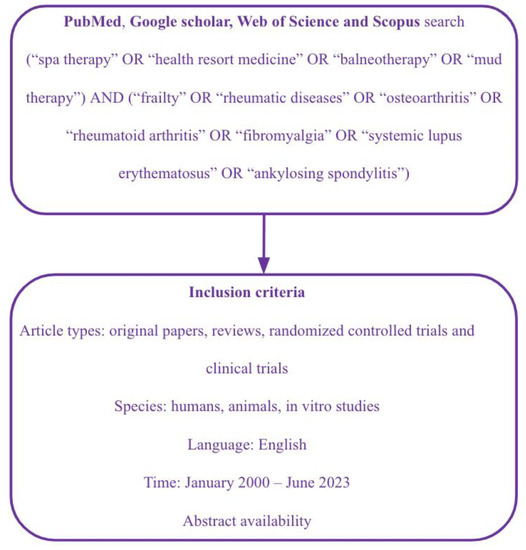
3. Frailty in Rheumatic Diseases
4. Spa Therapy Effects on Frailty in Rheumatic Diseases: Immunological Aspects
5. Spa Therapy Effects on Frailty in Rheumatic Diseases: Clinical Evidence
5.1. Spa Therapy for OA Patients
5.2. Preventive and Rehabilitative Strategies in the Spa Setting for OA Patients
5.3. Spa Therapy for RA Patients
5.4. Preventive and Rehabilitative Strategies in the Spa Setting for RA Patients
5.5. Spa Therapy for FM Patients
5.6. Preventive and Rehabilitative Strategies in the Spa Setting for FM Patients
5.7. Spa Therapy for SLE Patients
5.8. Other Effects

6. Discussion
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kojima, G.; Liljas, A.E.; Iliffe, S. Frailty syndrome: Implications and challenges for health care policy. Risk Manag. Health Policy 2019, 12, 23–30. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Morley, J.E. Frailty and sarcopenia in elderly. Wien. Klin. Wochenschr. 2016, 128, 439–445. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in Older adults: Evidence for a phenotype. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef] [PubMed]
- Fried, L.P.; Ferrucci, L.; Darer, J.; Williamson, J.D.; Anderson, G. Untangling the Concepts of Disability, Frailty, and Comorbidity: Implications for Improved Targeting and Care. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2004, 59, M255–M263. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Motta, F.; Sica, A.; Selmi, C. Frailty in Rheumatic Diseases. Front. Immunol. 2020, 11, 576134. [Google Scholar] [CrossRef] [PubMed]
- Kahn, A.; Fraga, M.F. Epigenetics and Aging: Status, Challenges, and Needs for the Future. J. Gerontol. Ser. A 2009, 64, 195–198. [Google Scholar] [CrossRef][Green Version]
- Alberro, A.; Iribarren-Lopez, A.; Sáenz-Cuesta, M.; Matheu, A.; Vergara, I.; Otaegui, D. Inflammaging markers characteristic of advanced age show similar levels with frailty and dependency. Sci. Rep. 2021, 11, 4358. [Google Scholar] [CrossRef]
- Gutenbrunner, C.; Bender, T.; Cantista, P.; Karagülle, Z. A proposal for a worldwide definition of health resort medicine, balneology, medical hydrology and climatology. Int. J. Biometeorol. 2010, 54, 495–507. [Google Scholar] [CrossRef]
- Kocyigit, B.F. Spa therapy: A geriatric perspective. Anti-Aging East. Eur. 2023, 2, 45–48. [Google Scholar] [CrossRef]
- Masiero, S.; Maccarone, M.C. Health resort therapy interventions in the COVID-19 pandemic era: What next? Int. J. Biometeorol. 2021, 65, 1995–1997. [Google Scholar] [CrossRef]
- Maccarone, M.C.; Masiero, S. Spa therapy interventions for post respiratory rehabilitation in COVID-19 subjects: Does the review of recent evidence suggest a role? Environ. Sci. Pollut. Res. 2021, 28, 46063–46066. [Google Scholar] [CrossRef]
- Bernetti, A.; Mangone, M.; Alviti, F.; Paolucci, T.; Attanasi, C.; Murgia, M.; Di Sante, L.; Agostini, F.; Vitale, M.; Paoloni, M. Spa therapy and rehabilitation of musculoskeletal pathologies: A proposal for best practice in Italy. Int. J. Biometeorol. 2020, 64, 905–914. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, A. Foreword: Balneotherapy in rheumatic diseases. Int. J. Biometeorol. 2020, 64, 903–904. [Google Scholar] [CrossRef] [PubMed]
- Maccarone, M.C.; Magro, G.; Solimene, U.; Scanu, A.; Masiero, S. From in vitro research to real life studies: An extensive narrative review of the effects of balneotherapy on human immune response. Sport Sci. Health 2021, 17, 817–835. [Google Scholar] [CrossRef] [PubMed]
- Cheleschi, S.; Tenti, S.; Seccafico, I.; Gálvez, I.; Fioravanti, A.; Ortega, E. Balneotherapy year in review 2021: Focus on the mechanisms of action of balneotherapy in rheumatic diseases. Environ. Sci. Pollut. Res. 2022, 29, 8054–8073. [Google Scholar] [CrossRef]
- Tognolo, L.; Coraci, D.; Fioravanti, A.; Tenti, S.; Scanu, A.; Magro, G.; Maccarone, M.C.; Masiero, S. Clinical Impact of Balneotherapy and Therapeutic Exercise in Rheumatic Diseases: A Lexical Analysis and Scoping Review. Appl. Sci. 2022, 12, 7379. [Google Scholar] [CrossRef]
- Fazaa, A.; Souabni, L.; Ben Abdelghani, K.; Kassab, S.; Chekili, S.; Zouari, B.; Hajri, R.; Laatar, A.; Zakraoui, L. Comparison of the clinical effectiveness of thermal cure and rehabilitation in knee osteoarthritis. A randomized therapeutic trial. Ann. Phys. Rehabil. Med. 2014, 57, 561–569. [Google Scholar] [CrossRef][Green Version]
- Altan, L.; Bingöl, Ü.; Aslan, M.; Yurtkuran, M. The effect of balneotherapy on patients with ankylosing spondylitis. Scand. J. Rheumatol. 2006, 35, 283–289. [Google Scholar] [CrossRef]
- Thompson, E.J.; Williams, D.M.; Walker, A.J.; Mitchell, R.E.; Niedzwiedz, C.L.; Yang, T.C.; Huggins, C.F.; Kwong, A.S.F.; Silverwood, R.J.; Di Gessa, G.; et al. Long COVID burden and risk factors in 10 UK longitudinal studies and electronic health records. Nat. Commun. 2022, 13, 3528. [Google Scholar] [CrossRef]
- Costantino, M.; Conti, V.; Corbi, G.; Ciancarelli, I.; Morone, G.; Filippelli, A. Sulphurous Crenotherapy Is Effective at Reducing Pain and Disability in Overweight/Obese Patients Affected by Chronic Low Back Pain from Spine Osteoarthritis. Healthcare 2022, 10, 1800. [Google Scholar] [CrossRef]
- Tamás, B.; Gabriella, K.; Kristóf, A.; Anett, I.; Pál, K.J.; Bálint, T.; Péter, L.; Márton, P.; Katalin, N. The Effects of Lakitelek Thermal Water and Tap Water on Skin Microbiome, a Randomized Control Pilot Study. Life 2023, 13, 746. [Google Scholar] [CrossRef] [PubMed]
- Van Tubergen, A.; Landewé, R.; Van Der Heijde, D.; Hidding, A.; Wolter, N.; Asscher, M.; Falkenbach, A.; Genth, E.; Goei Thè, H.; van der Linden, S. Combined spa–exercise therapy is effective in patients with ankylosing spondylitis: A randomized controlled trial. Arthritis Rheum. 2001, 45, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Arksey, H.; O’Malley, L. Scoping Studies: Advancing the Methodology. Int. J. Soc. Res. Methodol. 2005, 8, 19–32. [Google Scholar] [CrossRef][Green Version]
- Levac, D.; Colquhoun, H.; O’Brien, K.K. Scoping studies: Advancing the methodology. Implement. Sci. 2010, 5, 69. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chen, X.; Mao, G.; Leng, S.X. Frailty syndrome: An overview. Clin. Interv. Aging 2014, 9, 433–441. [Google Scholar] [CrossRef][Green Version]
- Howlett, S.E.; Rutenberg, A.D.; Rockwood, K. The degree of frailty as a translational measure of health in aging. Nat. Aging 2021, 1, 651–665. [Google Scholar] [CrossRef]
- Bisset, E.S.; Howlett, S.E. The biology of frailty in humans and animals: Understanding frailty and promoting translation. Aging Med. 2019, 2, 27–34. [Google Scholar] [CrossRef]
- Fuggle, N.; Laskou, F.; Harvey, N.; Dennison, E. A review of epigenetics and its association with ageing of muscle and bone. Maturitas 2022, 165, 12–17. [Google Scholar] [CrossRef]
- Hamerman, D. Toward an Understanding of Frailty. Ann. Intern. Med. 1999, 130, 945–950. [Google Scholar] [CrossRef]
- Van Epps, P.; Oswald, D.; Higgins, P.A.; Hornick, T.R.; Aung, H.; Banks, R.E.; Wilson, B.M.; Burant, C.; Gravenstein, S.; Canaday, D.H. Frailty has a stronger association with inflammation than age in older veterans. Immun. Ageing 2016, 13, 1–9. [Google Scholar] [CrossRef][Green Version]
- Franceschi, C.; Bonafe, M.; Valensin, S.; Olivieri, F.; De Luca, M.; Ottaviani, E.; De Benedictis, G. Inflamm-aging: An evolutionary perspective on immunosenescence. Ann. N. Y. Acad. Sci. 2000, 908, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Carraro, U.; Marcante, A.; Ravara, B.; Albertin, G.; Maccarone, M.C.; Piccione, F.; Kern, H.; Masiero, S. Skeletal muscle weakness in older adults home-restricted due to COVID-19 pandemic: A role for full-body in-bed gym and functional electrical stimulation. Aging Clin. Exp. Res. 2021, 33, 2053–2059. [Google Scholar] [CrossRef] [PubMed]
- Carraro, U.; Gava, K.; Baba, A.; Marcante, A.; Piccione, F. To Contrast and Reverse Skeletal Muscle Atrophy by Full-Body In-Bed Gym, a Mandatory Lifestyle for Older Olds and Borderline Mobility-Impaired Persons. Muscle Atrophy 2018, 1088, 549–560. [Google Scholar] [CrossRef]
- Leng, S.X.; Xue, Q.-L.; Tian, J.; Walston, J.D.; Fried, L.P. Inflammation and Frailty in Older Women. J. Am. Geriatr. Soc. 2007, 55, 864–871. [Google Scholar] [CrossRef] [PubMed]
- Leng, S.X.; Xue, Q.-L.; Tian, J.; Huang, Y.; Yeh, S.-H.; Fried, L.P. Associations of neutrophil and monocyte counts with frailty in community-dwelling disabled older women: Results from the Women’s Health and Aging Studies I. Exp. Gerontol. 2009, 44, 511–516. [Google Scholar] [CrossRef]
- Semba, R.D.; Margolick, J.B.; Leng, S.; Walston, J.; Ricks, M.O.; Fried, L.P. T cell subsets and mortality in older community-dwelling women. Exp. Gerontol. 2005, 40, 81–87. [Google Scholar] [CrossRef]
- Puts, M.T.E.; Visser, M.; Twisk, J.W.R.; Deeg, D.J.H.; Lips, P. Endocrine and inflammatory markers as predictors of frailty. Clin. Endocrinol. 2005, 63, 403–411. [Google Scholar] [CrossRef][Green Version]
- Buchmann, N.; Fielitz, J.; Spira, D.; König, M.; Norman, K.; Pawelec, G.; Goldeck, D.; Demuth, I.; Steinhagen-Thiessen, E. Muscle Mass and Inflammation in Older Adults: Impact of the Metabolic Syndrome. Gerontology 2022, 68, 989–998. [Google Scholar] [CrossRef]
- Soysal, P.; Stubbs, B.; Lucato, P.; Luchini, C.; Solmi, M.; Peluso, R.; Sergi, G.; Isik, A.T.; Manzato, E.; Maggi, S.; et al. Inflammation and frailty in the elderly: A systematic review and meta-analysis. Ageing Res. Rev. 2016, 31, 1–8. [Google Scholar] [CrossRef]
- Ershler, W.B.; Keller, E.T. Age-Associated Increased Interleukin-6 Gene Expression, Late-Life Diseases, and Frailty. Annu. Rev. Med. 2000, 51, 245–270. [Google Scholar] [CrossRef]
- Halil, M.; Kizilarslanoglu, M.C.; Kuyumcu, M.E.; Yesil, Y.; Jentoft, A.J.C. Cognitive aspects of frailty: Mechanisms behind the link between frailty and cognitive impairment. J. Nutr. Health Aging 2015, 19, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Michaud, M.; Balardy, L.; Moulis, G.; Gaudin, C.; Peyrot, C.; Vellas, B.; Cesari, M.; Nourhashemi, F. Proinflammatory Cytokines, Aging, and Age-Related Diseases. J. Am. Med. Dir. Assoc. 2013, 14, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Leng, S.X.; Yang, H.; Walston, J.D. Decreased cell proliferation and altered cytokine production in frail older adults. Aging Clin. Exp. Res. 2004, 16, 249–252. [Google Scholar] [CrossRef] [PubMed]
- Biggelaar, A.H.v.D.; Huizinga, T.W.; de Craen, A.J.; Gussekloo, J.; Heijmans, B.T.; Frölich, M.; Westendorp, R.G. Impaired innate immunity predicts frailty in old age. The Leiden 85-plus study. Exp. Gerontol. 2004, 39, 1407–1414. [Google Scholar] [CrossRef]
- Walston, J.; Fedarko, N.; Yang, H.; Leng, S.; Beamer, B.; Espinoza, S.; Lipton, A.; Zheng, H.; Becker, K. The Physical and Biological Characterization of a Frail Mouse Model. J. Gerontol. Ser. A 2008, 63, 391–398. [Google Scholar] [CrossRef][Green Version]
- Lorenzi, M.; Lorenzi, T.; Marzetti, E.; Landi, F.; Vetrano, D.L.; Settanni, S.; Antocicco, M.; Bonassi, S.; Valdiglesias, V.; Bernabei, R.; et al. Association of frailty with the serine protease HtrA1 in older adults. Exp. Gerontol. 2016, 81, 8–12. [Google Scholar] [CrossRef]
- Carrieri, G.; Marzi, E.; Olivieri, F.; Marchegiani, F.; Cavallone, L.; Cardelli, M.; Giovagnetti, S.; Stecconi, R.; Molendini, C.; Trapassi, C.; et al. The G/C915 polymorphism of transforming growth factor β1 is associated with human longevity: A study in Italian centenarians. Aging Cell 2004, 3, 443–448. [Google Scholar] [CrossRef]
- Scanzello, C.R.; Loeser, R.F. Editorial: Inflammatory Activity in Symptomatic Knee Osteoarthritis: Not All Inflammation Is Local. Arthritis Rheumatol. 2015, 67, 2797–2800. [Google Scholar] [CrossRef][Green Version]
- Chow, Y.Y.; Chin, K.-Y. The Role of Inflammation in the Pathogenesis of Osteoarthritis. Mediat. Inflamm. 2020, 2020, 1–19. [Google Scholar] [CrossRef][Green Version]
- Tognolo, L.; Maccarone, M.C.; De Trane, S.; Scanu, A.; Masiero, S.; Fiore, P. Therapeutic Exercise and Conservative Injection Treatment for Early Knee Osteoarthritis in Athletes: A Scoping Review. Medicina 2022, 58, 69. [Google Scholar] [CrossRef]
- Punzi, L.; Galozzi, P.; Luisetto, R.; Favero, M.; Ramonda, R.; Oliviero, F.; Scanu, A. Post-traumatic arthritis: Overview on pathogenic mechanisms and role of inflammation. RMD Open 2016, 2, e000279. [Google Scholar] [CrossRef] [PubMed]
- Livshits, G.; Zhai, G.; Hart, D.J.; Kato, B.S.; Wang, H.; Williams, F.M.K.; Spector, T.D. Interleukin-6 is a significant predictor of radiographic knee osteoarthritis: The Chingford study. Arthritis Rheum. 2009, 60, 2037–2045. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Spector, T.D.; Hart, D.J.; Nandra, D.; Doyle, D.V.; Mackillop, N.; Gallimore, J.R.; Pepys, M.B. Low-level increases in serum C-reactive protein are present in early osteoarthritis of the knee and predict progressive disease. Arthritis Rheum. 1997, 40, 723–727. [Google Scholar] [CrossRef] [PubMed]
- Wise, B.L.; Parimi, N.; Zhang, Y.; Cawthon, P.M.; Barrett-Connor, E.; Ensrud, K.; Lane, N.E.; The Osteoporotic Fractures in Men (MrOS) Group. Frailty and Hip Osteoarthritis in Men in the MrOS Cohort. J. Gerontol. Ser. A 2014, 69, 602–608. [Google Scholar] [CrossRef] [PubMed][Green Version]
- O’brien, M.S.; McDougall, J.J. Age and frailty as risk factors for the development of osteoarthritis. Mech. Ageing Dev. 2019, 180, 21–28. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef][Green Version]
- Pegreffi, F.; Balestra, A.; De Lucia, O.; Smith, L.; Barbagallo, M.; Veronese, N. Prevalence of Sarcopenia in Knee Osteoarthritis: A Systematic Review and Meta-Analysis. J. Clin. Med. 2023, 12, 1532. [Google Scholar] [CrossRef]
- Salaffi, F.; Di Matteo, A.; Farah, S.; Di Carlo, M. Inflammaging and Frailty in Immune-Mediated Rheumatic Diseases: How to Address and Score the Issue. Clin. Rev. Allergy Immunol. 2022, 64, 206–221. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Romero-Yuste, S.; Carmona, E.C.; Nolla, J.M. Sarcopenia, immune-mediated rheumatic diseases, and nutritional interventions. Aging Clin. Exp. Res. 2021, 33, 2929–2939. [Google Scholar] [CrossRef]
- Torii, M.; Hashimoto, M.; Hanai, A.; Fujii, T.; Furu, M.; Ito, H.; Uozumi, R.; Hamaguchi, M.; Terao, C.; Yamamoto, W.; et al. Prevalence and factors associated with sarcopenia in patients with rheumatoid arthritis. Mod. Rheumatol. 2019, 29, 589–595. [Google Scholar] [CrossRef]
- Katz, P. Fatigue in Rheumatoid Arthritis. Curr. Rheumatol. Rep. 2017, 19, 25. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.; Jackson, T.; Sapey, E.; Lord, J.M. Frailty and sarcopenia: The potential role of an aged immune system. Ageing Res. Rev. 2017, 36, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Velilla, N.; Fernández-Solà, J.; Santaeugènia, S.; Mas, M. Geriatric frailty applied to fibromyalgia patients. Rheumatol. Int. 2015, 35, 193–194. [Google Scholar] [CrossRef]
- Lieber, S.B.; Nahid, M.; Paget, S.; Berman, J.R.; Barbhaiya, M.; Sammaritano, L.R.; Kirou, K.; Carrino, J.A.; Rajan, M.; Sheira, D.; et al. Evaluation of a Patient-reported Frailty Tool in Women With Systemic Lupus Erythematosus. J. Rheumatol. 2022, 49, 60–67. [Google Scholar] [CrossRef]
- Ensrud, K.E.; Blackwell Ma, T.L.; Cauley, J.A.; Cummings, S.R.; Barrett-Connor, E.; Dam, T.-T.L.; Hoffman, A.R.; Shikany, J.M.; Lane, N.E.; Stefanick, M.L.; et al. Circulating 25-Hydroxyvitamin D Levels and Frailty in Older Men: The Osteoporotic Fractures in Men Study. J. Am. Geriatr. Soc. 2011, 59, 101–106. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shardell, M.; Hicks, G.E.; Miller, R.R.; Kritchevsky, S.; Andersen, D.; Bandinelli, S.; Cherubini, A.; Ferrucci, L. Association of Low Vitamin D Levels With the Frailty Syndrome in Men and Women. J. Gerontol. Ser. A 2009, 64A, 69–75. [Google Scholar] [CrossRef][Green Version]
- Maccarone, M.C.; Masiero, S.; Papathanasiou, J.; Panayotov, K.; Kashilskah, Y.; Prokopidis, K.; Papanastasiou, C.; Tyllianakis, M.; Dionyssiotis, Y. Frailty education: Promoting geriatric competencies among Physical Medicine and Rehabilitation residents. Am. J. Phys. Med. Rehabil. 2023. [Google Scholar] [CrossRef]
- Choi, M.; Kim, H.; Bae, J. Correction: Does the combination of resistance training and a nutritional intervention have a synergic effect on muscle mass, strength, and physical function in older adults? A systematic review and meta-analysis. BMC Geriatr. 2022, 22, 531. [Google Scholar] [CrossRef]
- Masiero, S. Thermal rehabilitation and osteoarticular diseases of the elderly. Aging Clin. Exp. Res. 2008, 20, 189–194. [Google Scholar] [CrossRef]
- Masiero, S.; Vittadini, F.; Ferroni, C.; Bosco, A.; Serra, R.; Frigo, A.C.; Frizziero, A. The role of thermal balneotherapy in the treatment of obese patient with knee osteoarthritis. Int. J. Biometeorol. 2018, 62, 243–252. [Google Scholar] [CrossRef]
- Cozzi, F.; Ciprian, L.; Carrara, M.; Galozzi, P.; Zanatta, E.; Scanu, A.; Sfriso, P.; Punzi, L. Balneotherapy in chronic inflammatory rheumatic diseases—A narrative review. Int. J. Biometeorol. 2018, 62, 2065–2071. [Google Scholar] [CrossRef] [PubMed]
- McAlindon, T.E.; Bannuru, R.R.; Sullivan, M.C.; Arden, N.K.; Berenbaum, F.; Bierma-Zeinstra, S.M.; Hawker, G.A.; Henrotin, Y.; Hunter, D.J.; Kawaguchi, H.; et al. OARSI guidelines for the non-surgical management of knee osteoarthritis. Osteoarthr. Cartil. 2014, 22, 363–388. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Scanu, A.; Tognolo, L.; Maccarone, M.C.; Masiero, S. Immunological Events, Emerging Pharmaceutical Treatments and Therapeutic Potential of Balneotherapy on Osteoarthritis. Front. Pharmacol. 2021, 12, 681871. [Google Scholar] [CrossRef] [PubMed]
- Cheleschi, S.; Gallo, I.; Tenti, S. A comprehensive analysis to understand the mechanism of action of balneotherapy: Why, how, and where they can be used? Evidence from in vitro studies performed on human and animal samples. Int. J. Biometeorol. 2020, 64, 1247–1261. [Google Scholar] [CrossRef]
- Ortega, E.; Gálvez, I.; Hinchado, M.D.; Guerrero, J.; Martín-Cordero, L.; Torres-Piles, S. Anti-inflammatory effect as a mechanism of effectiveness underlying the clinical benefits of pelotherapy in osteoarthritis patients: Regulation of the altered inflammatory and stress feedback response. Int. J. Biometeorol. 2017, 61, 1777–1785. [Google Scholar] [CrossRef]
- Gálvez, I.; Torres-Piles, S.; Ortega-Rincón, E. Balneotherapy, Immune System, and Stress Response: A Hormetic Strategy? Int. J. Mol. Sci. 2018, 19, 1687. [Google Scholar] [CrossRef][Green Version]
- Giannitti, C.; De Palma, A.; Pascarelli, N.A.; Cheleschi, S.; Giordano, N.; Galeazzi, M.; Fioravanti, A. Can balneotherapy modify microRNA expression levels in osteoarthritis? A comparative study in patients with knee osteoarthritis. Int. J. Biometeorol. 2017, 61, 2153–2158. [Google Scholar] [CrossRef]
- Fioravanti, A.; Cantarini, L.; Guidelli, G.M.; Galeazzi, M. Mechanisms of action of spa therapies in rheumatic diseases: What scientific evidence is there? Rheumatol. Int. 2011, 31, 1313–1321. [Google Scholar] [CrossRef]
- Teległów, A.; Seremak, J.; Golec, J.; Marchewka, J.; Golec, P.; Marchewka, U.; Maciejczyk, M.; Golec, E. The effect of sulfur baths on hemorheological properties of blood in patients with osteoarthritis. Sci. Rep. 2023, 13, 7960. [Google Scholar] [CrossRef]
- Buerck, J.P.; Burke, D.K.; Schmidtke, D.W.; Snyder, T.A.; Papavassiliou, D.; O’rear, E.A. A Flow Induced Autoimmune Response and Accelerated Senescence of Red Blood Cells in Cardiovascular Devices. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef][Green Version]
- Cozzi, F.; Carrara, M.; Sfriso, P.; Todesco, S.; Cima, L. Anti-inflammatory effect of mud-bath applications on adjuvant arthritis in rats. Clin. Exp. Rheumatol. 2004, 22, 763–766. [Google Scholar] [PubMed]
- Britschka, Z.M.N.; Teodoro, W.R.; Velosa, A.P.P.; de Mello, S.B.V. The efficacy of Brazilian black mud treatment in chronic experimental arthritis. Rheumatol. Int. 2007, 28, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Yücesoy, H.; Dönmez, A.; Atmaca-Aydın, E.; Yentür, S.P.; Saruhan-Direskeneli, G.; Ankaralı, H.; Erdoğan, N.; Karagülle, M.Z. Effects of balneological outpatient treatment on clinical parameters and serum cytokine levels in patients with chronic low back pain: A single-blind randomized controlled trial. Int. J. Biometeorol. 2021, 65, 1367–1376. [Google Scholar] [CrossRef]
- Angioni, M.; Denotti, A.; Pinna, S.; Sanna, C.; Montisci, F.; Dessole, G.; Loi, A.; Cauli, A. Spa therapy induces clinical improvement and protein changes in patients with chronic back pain. Reumatismo 2019, 71, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Tarner, I.H.; Müller-Ladner, U.; Uhlemann, C.; Lange, U. The effect of mild whole-body hyperthermia on systemic levels of TNF-alpha, IL-1beta, and IL-6 in patients with ankylosing spondylitis. Clin. Rheumatol. 2009, 28, 397–402. [Google Scholar] [CrossRef]
- Shehata, M.; Schwarzmeier, J.D.; Hilgarth, M.; Demirtas, D.; Richter, D.; Hubmann, R.; Boeck, P.; Leiner, G.; Falkenbach, A. Effect of combined spa-exercise therapy on circulating TGF-β1 levels in patients with ankylosing spondylitis. Wien. Klin. Wochenschr. 2006, 118, 266–272. [Google Scholar] [CrossRef]
- Ardıç, F.; Özgen, M.; Aybek, H.; Rota, S.; Çubukçu, D.; Gökgöz, A. Effects of balneotherapy on serum IL-1, PGE2 and LTB4 levels in fibromyalgia patients. Rheumatol. Int. 2007, 27, 441–446. [Google Scholar] [CrossRef]
- Maeda, T.; Kudo, Y.; Horiuchi, T.; Makino, N. Clinical and anti-aging effect of mud-bathing therapy for patients with fibromyalgia. Mol. Cell. Biochem. 2018, 444, 87–92. [Google Scholar] [CrossRef]
- Semiz, E.A.; Hizmetli, S.; Semiz, M.; Karadağ, A.; Adalı, M.; Tuncay, M.S.; Alim, B.; Hayta, E.; Uslu, A.U. Serum cortisol and dehydroepiandrosterone-sulfate levels after balneotherapy and physical therapy in patients with fibromyalgia. Saudi Med. J. 2016, 37, 544–550. [Google Scholar] [CrossRef]
- Živná, H.; Maric, L.; Gradošová, I.; Švejkovská, K.; Hubená, S.; Živný, P. The Effect of Mud-Bath Therapy on Bone Status in Rats During Adjuvant Subchronic Arthritis. Acta Med. 2012, 55, 133–137. [Google Scholar] [CrossRef][Green Version]
- Fioravanti, A.; Giannitti, C.; Cheleschi, S.; Simpatico, A.; Pascarelli, N.A.; Galeazzi, M. Circulating levels of adiponectin, resistin, and visfatin after mud-bath therapy in patients with bilateral knee osteoarthritis. Int. J. Biometeorol. 2015, 59, 1691–1700. [Google Scholar] [CrossRef]
- Koçak, F.A.; Kurt, E.E.; Sezgin, F.M.; Şaş, S.; Tuncay, F.; Erdem, H.R. The effect of balneotherapy on body mass index, adipokine levels, sleep disturbances, and quality of life of women with morbid obesity. Int. J. Biometeorol. 2020, 64, 1463–1472. [Google Scholar] [CrossRef] [PubMed]
- Kamioka, H.; Nakamura, Y.; Okada, S.; Kitayuguchi, J.; Kamada, M.; Honda, T.; Matsui, Y.; Mutoh, Y. Effectiveness of Comprehensive Health Education Combining Lifestyle Education and Hot Spa Bathing for Male White-Collar Employees: A Randomized Controlled Trial with 1-Year Follow-Up. J. Epidemiol. 2009, 19, 219–230. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hanzel, A.; Horvát, K.; Molics, B.; Berényi, K.; Nemeth, B.; Szendi, K.; Varga, C. Clinical improvement of patients with osteoarthritis using thermal mineral water at Szigetvár Spa—Results of a randomised double-blind controlled study. Int. J. Biometeorol. 2018, 62, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Szakál, E.; Szántó, D.; Szekeres, L.; Bender, T. Evaluation of the effect of Lake Hévíz thermal mineral water in patients with osteoarthritis of the knee: A randomized, controlled, single-blind, follow-up study. Eur. J. Phys. Rehabil. Med. 2014, 50, 373–381. [Google Scholar]
- Annegret, F.; Thomas, F. Long-term benefits of radon spa therapy in rheumatic diseases: Results of the randomised, multi-centre IMuRa trial. Rheumatol. Int. 2013, 33, 2839–2850. [Google Scholar] [CrossRef]
- Branco, M.; Rêgo, N.N.; Silva, P.H.; Archanjo, I.E.; Ribeiro, M.C.; Trevisani, V.F. Bath thermal waters in the treatment of knee osteoarthritis: A randomized controlled clinical trial. Eur. J. Phys. Rehabil. Med. 2016, 52, 422–430. [Google Scholar]
- Fioravanti, A.; Giannitti, C.; Bellisai, B.; Iacoponi, F.; Galeazzi, M. Efficacy of balneotherapy on pain, function and quality of life in patients with osteoarthritis of the knee. Int. J. Biometeorol. 2012, 56, 583–590. [Google Scholar] [CrossRef]
- Kovacs, I.; Bender, T. The therapeutic effects of Cserkeszölö thermal water in osteoarthritis of the knee: A double blind, controlled, follow-up study. Rheumatol. Int. 2002, 21, 218–221. [Google Scholar] [CrossRef]
- Sherman, G.; Zeller, L.; Avriel, A.; Friger, M.; Harari, M.; Sukenik, S. Intermittent balneotherapy at the Dead Sea area for patients with knee osteoarthritis. Isr. Med. Assoc. J. IMAJ 2009, 11, 88. [Google Scholar]
- Kovács, C.; Pecze, M.; Tihanyi, Á.; Kovács, L.; Balogh, S.; Bender, T. The effect of sulphurous water in patients with osteoarthritis of hand. Double-blind, randomized, controlled follow-up study. Clin. Rheumatol. 2012, 31, 1437–1442. [Google Scholar] [CrossRef] [PubMed]
- Zwolińska, J.; Gąsior, M. Effects of complex spa therapy in patients with osteoarthritis of the spine receiving treatments in health resorts in south-eastern Poland. Sci. Rep. 2022, 12, 14663. [Google Scholar] [CrossRef] [PubMed]
- Protano, C.; Fontana, M.; De Giorgi, A.; Marotta, D.; Cocomello, N.; Crucianelli, S.; Del Cimmuto, A.; Vitali, M. Balneotherapy for osteoarthritis: A systematic review. Rheumatol. Int. 2023. [Google Scholar] [CrossRef]
- Antúnez, L.E.; Puértolas, B.C.; Burgos, B.I.; Porto-Payán, J.M.; Piles, S.T.T. Efectos de la terapia con peloides sobre el dolor percibido y la calidad de vida relacionada con la salud en pacientes con artrosis de rodilla. Reumatol. Clínica 2013, 9, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Sarsan, A.; Akkaya, N.; Özgen, M.; Yildiz, N.; Atalay, N.S.; Ardic, F. Comparing the efficacy of mature mud pack and hot pack treatments for knee osteoarthritis. J. Back Musculoskelet. Rehabil. 2012, 25, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Güngen, G.; Ardic, F.; Fιndıkoğlu, G.; Rota, S. The effect of mud pack therapy on serum YKL-40 and hsCRP levels in patients with knee osteoarthritis. Rheumatol. Int. 2012, 32, 1235–1244. [Google Scholar] [CrossRef] [PubMed]
- Király, M.; Kővári, E.; Hodosi, K.; Bálint, P.V.; Bender, T. The effects of Tiszasüly and Kolop mud pack therapy on knee osteoarthritis: A double-blind, randomised, non-inferiority controlled study. Int. J. Biometeorol. 2020, 64, 943–950. [Google Scholar] [CrossRef][Green Version]
- Tefner, I.K.; Gaál, R.; Koroknai, A.; Ráthonyi, A.; Gáti, T.; Monduk, P.; Kiss, E.; Kovács, C.; Bálint, G.; Bender, T. The effect of Neydharting mud-pack therapy on knee osteoarthritis: A randomized, controlled, double-blind follow-up pilot study. Rheumatol. Int. 2013, 33, 2569–2576. [Google Scholar] [CrossRef]
- Odabasi, E.; Turan, M.; Erdem, H.; Tekbas, F. Does Mud Pack Treatment Have Any Chemical Effect? A Randomized Controlled Clinical Study. J. Altern. Complement. Med. 2008, 14, 559–565. [Google Scholar] [CrossRef]
- D’angelo, D.; Coclite, D.; Napoletano, A.; Fauci, A.J.; Latina, R.; Gianola, S.; Castellini, G.; Salomone, K.; Gambalunga, F.; Sperati, F.; et al. The efficacy of balneotherapy, mud therapy and spa therapy in patients with osteoarthritis: An overview of reviews. Int. J. Biometeorol. 2021, 65, 1255–1271. [Google Scholar] [CrossRef]
- Antonelli, M.; Donelli, D.; Veronesi, L.; Vitale, M.; Pasquarella, C. Clinical efficacy of medical hydrology: An umbrella review. Int. J. Biometeorol. 2021, 65, 1597–1614. [Google Scholar] [CrossRef] [PubMed]
- Evcik, D.; Kavuncu, V.; Yeter, A.; Yigit, I. The efficacy of balneotherapy and mud-pack therapy in patients with knee osteoarthritis. Jt. Bone Spine 2007, 74, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Pascarelli, N.A.; Cheleschi, S.; Bacaro, G.; Guidelli, G.M.; Galeazzi, M.; Fioravanti, A. Effect of Mud-Bath Therapy on Se-rum Biomarkers in Patients with Knee Osteoarthritis: Results from a Randomized Controlled Trial. Isr. Med. Assoc. J. 2016, 18, 232–237. [Google Scholar]
- Wigler, I.; Elkayam, O.; Paran, D.; Yaron, M. Spa therapy for gonarthrosis: A prospective study. Rheumatol. Int. 1995, 15, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Mueller, J.; Langbein, T.; Mishra, A.; Baum, R.P. Safety of High-Dose Botulinum Toxin Injections for Parotid and Submandibular Gland Radioprotection. Toxins 2022, 14, 64. [Google Scholar] [CrossRef]
- Adıgüzel, T.; Arslan, B.; Gürdal, H.; Karagülle, M.Z. Evaluation of the therapeutic and the chemical effects of balneological treatment on clinical and laboratory parameters in knee osteoarthritis: A randomized, controlled, single-blinded trial. Int. J. Biometeorol. 2022, 66, 1257–1265. [Google Scholar] [CrossRef]
- Fioravanti, A.; Cantarini, L.; Bacarelli, M.R.; De Lalla, A.; Ceccatelli, L.; Blardi, P. Effects of Spa therapy on serum leptin and adiponectin levels in patients with knee osteoarthritis. Rheumatol. Int. 2011, 31, 879–882. [Google Scholar] [CrossRef][Green Version]
- Rat, A.-C.; Loeuille, D.; Vallata, A.; Bernard, L.; Spitz, E.; Desvignes, A.; Boulange, M.; Paysant, J.; Guillemin, F.; Chary-Valckenaere, I. Spa therapy with physical rehabilitation is an alternative to usual spa therapy protocol in symptomatic knee osteoarthritis. Sci. Rep. 2020, 10, 11004. [Google Scholar] [CrossRef]
- Kovács, C.; Bozsik, Á.; Pecze, M.; Borbély, I.; Fogarasi, A.; Kovács, L.; Tefner, I.K.; Bender, T. Effects of sulfur bath on hip osteoarthritis: A randomized, controlled, single-blind, follow-up trial: A pilot study. Int. J. Biometeorol. 2016, 60, 1675–1680. [Google Scholar] [CrossRef]
- Cantista, P.; Maraver, F. Balneotherapy for knee osteoarthritis in S. Jorge: A randomized controlled trial. Int. J. Biometeorol. 2020, 64, 1027–1038. [Google Scholar] [CrossRef]
- Varzaityte, L.; Kubilius, R.; Rapoliene, L.; Bartuseviciute, R.; Balcius, A.; Ramanauskas, K.; Nedzelskiene, I. The effect of balneotherapy and peloid therapy on changes in the functional state of patients with knee joint osteoarthritis: A randomized, controlled, single-blind pilot study. Int. J. Biometeorol. 2019, 64, 955–964. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Coraci, D.; Tognolo, L.; Maccarone, M.C.; Santilli, G.; Ronconi, G.; Masiero, S. Water-Based Rehabilitation in the Elderly: Data Science Approach to Support the Conduction of a Scoping Review. Appl. Sci. 2022, 12, 8999. [Google Scholar] [CrossRef]
- Maccarone, M.C.; Magro, G.; Albertin, C.; Barbetta, G.; Barone, S.; Castaldelli, C.; Manica, P.; Marcoli, S.; Mediati, M.; Minuto, D.; et al. Short-time effects of spa rehabilitation on pain, mood and quality of life among patients with degenerative or post-surgery musculoskeletal disorders. Int. J. Biometeorol. 2023, 67, 29–36. [Google Scholar] [CrossRef] [PubMed]
- van der Zee-Neuen, A.; Fuchs, J.; Wildburger, S.; Gaisberger, M.; Kloppenburg, M.; Fioravanti, A.; Stamm, T.; Offenbaecher, M.; Radlmueller, R.; Foisner, W.; et al. Improvement of Pain Symptoms in Musculoskeletal Diseases After Multimodal Spa Therapy in the Austrian Gastein Valley—A Study Based on Longitudinal Registry Data. Int. J. Public Health 2023, 68, 1605931. [Google Scholar] [CrossRef] [PubMed]
- Karagülle, M.; Kardeş, S.; Karagülle, O.; Dişçi, R.; Avcı, A.; Durak, I.; Karagülle, M.Z. Effect of spa therapy with saline balneotherapy on oxidant/antioxidant status in patients with rheumatoid arthritis: A single-blind randomized controlled trial. Int. J. Biometeorol. 2017, 61, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Güneri, F.D.; Forestier, F.B.E.; Forestier, R.J.; Karabulut, S.S.; Karaarslan, F.; Karagülle, M.Z.; Karagülle, M. Peloidotherapy in rheumatoid arthritis: A pilot randomized clinical trial. Int. J. Biometeorol. 2021, 65, 2171–2180. [Google Scholar] [CrossRef] [PubMed]
- Sukenik, S.; Buskila, D.; Neumann, L.; Kleiner-Baumgarten, A. Mud pack therapy in rheumatoid arthritis. Clin. Rheumatol. 1992, 11, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Sukenik, S.; Neumann, L.; Flusser, D.; Kleiner-Baumgarten, A.; Buskila, D. Balneotherapy for rheumatoid arthritis at the Dead Sea. Isr. J. Med. Sci. 1995, 31, 210–214. [Google Scholar]
- Fernandez-Gonzalez, M.; Fernandez-Lao, C.; Martin-Martin, L.; Gonzalez-Santos, A.; Lopez-Garzon, M.; Ortiz-Comino, L.; Lozano-Lozano, M. Therapeutic Benefits of Balneotherapy on Quality of Life of Patients with Rheumatoid Arthritis: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 13216. [Google Scholar] [CrossRef]
- Yurtkuran, M.; Yurtkuran, M.; Alp, A.; Nasırcılar, A.; Bingöl, Ü.; Altan, L.; Sarpdere, G. Balneotherapy and tap water therapy in the treatment of knee osteoarthritis. Rheumatol. Int. 2006, 27, 19–27. [Google Scholar] [CrossRef]
- De La Cruz, S.P. Influence of an Aquatic Therapy Program on Perceived Pain, Stress, and Quality of Life in Chronic Stroke Patients: A Randomized Trial. Int. J. Environ. Res. Public Health 2020, 17, 4796. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, A.; Manica, P.; Bortolotti, R.; Cevenini, G.; Tenti, S.; Paolazzi, G. Is balneotherapy effective for fibromyalgia? Results from a 6-month double-blind randomized clinical trial. Clin. Rheumatol. 2018, 37, 2203–2212. [Google Scholar] [CrossRef]
- Buskila, D.; Abu-Shakra, M.; Neumann, L.; Odes, L.; Shneider, E.; Flusser, D.; Sukenik, S. Balneotherapy for fibromyalgia at the Dead Sea. Rheumatol. Int. 2001, 20, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Dönmez, A.; Karagülle, M.Z.; Tercan, N.; Dinler, M.; Issever, H.; Turan, M.; Karagülle, M. SPA therapy in fibromyalgia: A randomised controlled clinic study. Rheumatol. Int. 2005, 26, 168–172. [Google Scholar] [CrossRef]
- Maindet, C.; Maire, A.; Vermorel, C.; Cracowski, C.; Rolland, C.; Forestier, R.; Comte, A.; Roques, C.-F.; Serra, E.; Bosson, J.-L. Spa Therapy for the Treatment of Fibromyalgia: An Open, Randomized Multicenter Trial. J. Pain 2021, 22, 940–951. [Google Scholar] [CrossRef]
- Jazani, A.M.; Azgomi, H.N.D.; Azgomi, A.N.D.; Ayati, M.H.; Azgomi, R.N.D. Efficacy of hydrotherapy, spa therapy, and balneotherapy on sleep quality: A systematic review. Int. J. Biometeorol. 2023, 67, 975–991. [Google Scholar] [CrossRef]
- Long, J.; Qin, Q.; Huang, Y.; Wang, T.; Jiang, J.; Gao, Q.; Chen, Y. Study on nondrug intervention of 7 days of balneotherapy combined with various sleep-promoting measures on people with sleep disorders: Preliminary and pilot study. Int. J. Biometeorol. 2023, 67, 457–464. [Google Scholar] [CrossRef]
- Castelli, L.; Galasso, L.; Mulè, A.; Ciorciari, A.; Fornasini, F.; Montaruli, A.; Roveda, E.; Esposito, F. Sleep and spa therapies: What is the role of balneotherapy associated with exercise? A systematic review. Front. Physiol. 2022, 13, 1560. [Google Scholar] [CrossRef]
- Ducamp, P.; Sichère, P.; Gayum, H.; Dubourg, K.; Roques, C.-F.; Journot, V. Therapeutic Patient Education for Fibromyalgia during Spa Therapy: The FiETT Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2022, 19, 4613. [Google Scholar] [CrossRef]
- Fritsch, K.; Nagy, G.; Szekanecz, Z.; Szűcs, G.; Kovacs, L.; Bender, T. Balneotherapy, a Complementary Non-pharmacological Approach for Non-Inflammatory Complaints in Systemic Lupus Erythematosus: A Pilot Study. Vivo 2022, 36, 3010–3017. [Google Scholar] [CrossRef] [PubMed]
- Maccarone, M.C.; Magro, G.; Tognolo, L.; Masiero, S. Post COVID-19 persistent fatigue: A proposal for rehabilitative interventions in the spa setting. Int. J. Biometeorol. 2021, 65, 2241–2243. [Google Scholar] [CrossRef]
- Maccarone, M.C.; Kamioka, H.; Cheleschi, S.; Tenti, S.; Masiero, S.; Kardeş, S. Italian and Japanese public attention toward balneotherapy in the COVID-19 era. Environ. Sci. Pollut. Res. 2021, 28, 61781–61789. [Google Scholar] [CrossRef]
- Masiero, S.; Maccarone, M.C.; Agostini, F. Health resort medicine can be a suitable setting to recover disabilities in patients tested negative for COVID-19 discharged from hospital? A challenge for the future. Int. J. Biometeorol. 2020, 64, 1807–1809. [Google Scholar] [CrossRef]
- Schnebelen-Berthier, C.; Negro, N.; Jaruga, A.; Roques, C.-F.; Lecerf, J.-M. Long term effect of spa therapy combined with patient education program on subjects with overweight and obesity—A controlled study. Obes. Res. Clin. Pract. 2019, 13, 492–498. [Google Scholar] [CrossRef]
- Fioravanti, A.; Adamczyk, P.; Pascarelli, N.A.; Giannitti, C.; Urso, R.; Tołodziecki, M.; Ponikowska, I. Clinical and biochemical effects of a 3-week program of diet combined with spa therapy in obese and diabetic patients: A pilot open study. Int. J. Biometeorol. 2015, 59, 783–789. [Google Scholar] [CrossRef] [PubMed]
- Kundakci, B.; Kaur, J.; Goh, S.L.; Hall, M.; Doherty, M.; Zhang, W.; Abhishek, A. Efficacy of nonpharmacological interventions for individual features of fibromyalgia: A systematic review and meta-analysis of randomised controlled trials. Pain 2022, 163, 1432–1445. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.-F.; Ma, K.-L.; Li, Q.-L.; Luan, F.-J.; Wang, Q.-B.; Zhang, M.-H.; Viswanath, O.; Myrcik, D.; Varrassi, G.; Wang, H.-Q. Balneotherapy for Fibromyalgia Syndrome: A Systematic Review and Meta-Analysis. J. Clin. Med. 2021, 10, 1493. [Google Scholar] [CrossRef] [PubMed]
- Bazzichi, L.; Da Valle, Y.; Rossi, A.; Giacomelli, C.; Sernissi, F.; Giannaccini, G.; Betti, L.; Ciregia, F.; Giusti, L.; Scarpellini, P.; et al. A multidisciplinary approach to study the effects of balneotherapy and mud-bath therapy treatments on fibromyalgia. Clin. Exp. Rheumatol. 2013, 31, S111–S120. [Google Scholar]
- Angulo, J.; El Assar, M.; Álvarez-Bustos, A.; Rodríguez-Mañas, L. Physical activity and exercise: Strategies to manage frailty. Redox Biol. 2020, 35, 101513. [Google Scholar] [CrossRef]
- Aguirre, L.E.; Villareal, D.T. Physical Exercise as Therapy for Frailty. Frailty Pathophysiol. Phenotype Patient Care 2015, 83, 83–92. [Google Scholar] [CrossRef][Green Version]
- Becker, B.E. Aquatic Therapy: Scientific Foundations and Clinical Rehabilitation Applications. PM&R 2009, 1, 859–872. [Google Scholar] [CrossRef]
- Maccarone, M.C.; Masiero, S. Can spa rehabilitative interventions play a role for patients suffering from neurodegenerative disorders at the early stages? A scoping review. Int. J. Biometeorol. 2022, 66, 2369–2377. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, A.; Karagülle, M.; Bender, T.; Karagülle, M.Z. Balneotherapy in osteoarthritis: Facts, fiction and gaps in knowledge. Eur. J. Integr. Med. 2017, 9, 148–150. [Google Scholar] [CrossRef]
- Masiero, S.; On behalf section of Rehabilitation in Environmental Thermal for Italian Society of Physical Medicine and Rehabilitation; Litwocenko, S.; Agostini, F. Rehabilitation in an Italian thermal setting: A new therapeutic strategy for patients with musculoskeletal disability—The results of an Italian survey. Int. J. Biometeorol. 2020, 64, 951–954. [Google Scholar] [CrossRef] [PubMed]
- Di Marco, R.; Pistonesi, F.; Cianci, V.; Biundo, R.; Weis, L.; Tognolo, L.; Baba, A.; Rubega, M.; Gentile, G.; Tedesco, C.; et al. Effect of Intensive Rehabilitation Program in Thermal Water on a Group of People with Parkinson’s Disease: A Retrospective Longitudinal Study. Healthcare 2022, 10, 368. [Google Scholar] [CrossRef] [PubMed]
- Masiero, S.; Maghini, I.; Mantovani, M.E.; Bakdounes, L.; Koutsikos, K.; Del Felice, A.; Sale, P. Is the aquatic thermal environment a suitable place for providing rehabilitative treatment for person with Parkinson’s disease? A retrospective study. Int. J. Biometeorol. 2019, 63, 13–18. [Google Scholar] [CrossRef]
- Grishechkina, I.A.; Lobanov, A.A.; Andronov, S.V.; Rachin, A.P.; Fesyun, A.D.; Ivanova, E.P.; Masiero, S.; Maccarone, M.C. Long-term outcomes of different rehabilitation programs in patients with long COVID syndrome: A cohort prospective study. Eur. J. Transl. Myol. 2023, 33. [Google Scholar] [CrossRef]
- Lobanov, A.A.; Grishechkina, I.A.; Andronov, S.V.; Barashkov, G.N.; Popov, A.I.; Fesyun, A.D.; Ivanova, E.P.; Maccarone, M.C.; Masiero, S. Can aquatic exercises contribute to the improvement of the gait stereotype function in patients with Long COVID outcomes? Eur. J. Transl. Myol. 2022, 32, 10698. [Google Scholar] [CrossRef]
- Antonelli, M.; Donelli, D. Effects of balneotherapy and spa therapy on levels of cortisol as a stress biomarker: A systematic review. Int. J. Biometeorol. 2018, 62, 913–924. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
|

